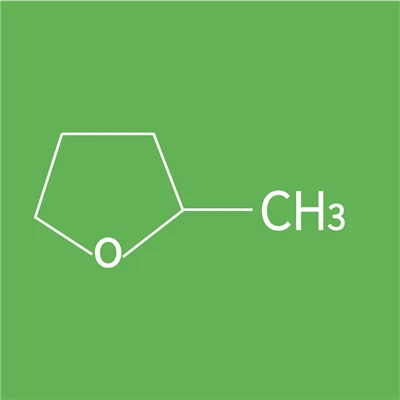
2-Methyltetrahydrofuran is primarily used as a high-boiling point alternative solvent to tetrahydrofuran (THF). It is also used as an electrolyte in secondary lithium batteries and as a component of alternative fuels. It is an important solvent for low-temperature reactions. 2-Methyltetrahydrofuran forms a glassy solid at low temperatures without crystallizing, making it a common solvent for spectroscopic studies at -196°C.
Another common use of 2-Methyltetrahydrofuran is as a solvent for Grignard reagents used in organometallic and two-phase chemical processes, as the oxygen atom in 2-Methyltetrahydrofuran can coordinate with the magnesium component of the Grignard reagent or the azeotropic drying product. 2-Methyltetrahydrofuran allows for a clean separation of the organic and aqueous phases, making it a common, but more expensive, alternative to THF.
2-Methyltetrahydrofuran is approved by the US Department of Energy as a gasoline additive. Furfural and other furan-based compounds (furfuryl alcohol, 2-methylfuran, tetrahydrofurfuryl alcohol) have a tendency to polymerize and are quite volatile. However, 2-methyltetrahydrofuran itself is more stable and less volatile, making it suitable for use as a motor fuel. 2-methyltetrahydrofuran has been promoted as an ecologically less hazardous alternative to tetrahydrofuran. [6] Although 2-methyltetrahydrofuran is more expensive, it can offer greater economies in the overall synthesis process. 2-methyltetrahydrofuran has solvent properties intermediate between those of diethyl ether and tetrahydrofuran, with limited water solubility and forming an azeotrope with water upon distillation. Its lower melting point allows it to be used in lower temperature reactions, while its higher boiling point relative to tetrahydrofuran allows for reflux at higher temperatures.